Introduction
Having thoroughly covered the difference between weight loss and fat loss and the importance of body composition, I would now like to build upon our discussion and talk about calories and energy balance given that these two topics are 1) widely misinterpreted, and 2) intimately involved in driving changes in body weight/composition—contrary to what some try to argue. Indeed, it is not hard to find claims that calories do not matter when it comes to weight gain or weight loss (and that other things such as hormones or just one single hormone is all that matters). This, however, is simply not an honest interpretation of the literature as will become evident shortly.
Because this is such a hefty topic, I will split it up into two parts (possibly three if needed). In Part 1 (today) I will cover the topic of energy and calories and begin to explain Energy Balance, at least from the energy intake side of the equation. In Part 2 I will explicitly cover energy expenditure and how measure it, as well as how the Energy Balance Equation changes in response to a variety of conditions (e.g. meal frequency, macronutrient composition, overfeeding and underfeeding) and the implication of such changes—it is here that I may need to break it up into three parts. (TBD!)
The main point in all of this is that calories do matter, energy balance is a thing, and all of it is more complicated than you realized or heard from some Instagram influencer.
Let us begin with the basics.
Energy
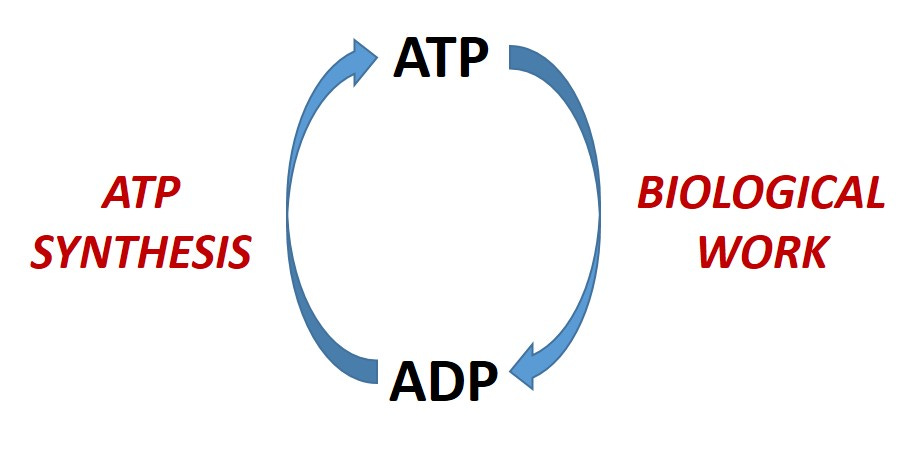
Energy is simply the capacity to do something—i.e. work. In your body, that means biological work that keeps you alive by maintaining ion gradients, building new macromolecules, allowing for locomotion, and so on and so forth. Should you stop doing any of this, you will die. (The fancy term is thermodynamic equilibrium.) Since energy cannot be created out of thin air, you need to obtain it (i.e. transfer it) from some other source—that source being food. Therefore, food contains energy and we put that food in our mouths and chemically digest it so that we can transfer the energy it contains (within its molecules) into the bonds of ATP (the universal cellular energy currency) that our cells constantly make that allows us to do work and stay alive.
This constant utilization and re-synthesis of ATP allows us to stave off entropy (the tendency for disorder)…for now. Should some of that energy be in excess of what is needed, it will be stored for later (as either fat or glycogen) and taken out of storage at the appropriate time. (Beach season, preferably!)
Calories: a primer
All of this begs the question: “How do we measure energy in food?” That is where calories come in. A Calorie (big ‘c’) is simply a unit of measure of energy. If you want to measure the distance between point A and point B, or the weight of an object, you could do that with a ruler or a scale, respectively. If you want to know the energy content of your food, let us say an apple that you are holding, you can do this by determining its Calorie content. Simple enough.
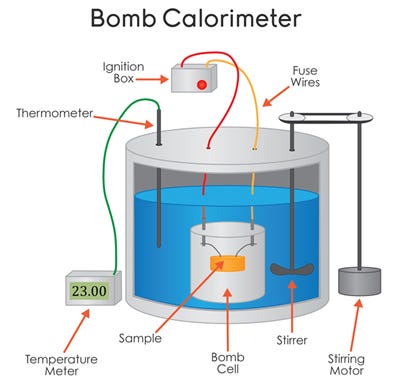
Getting a little deeper into the weeds, a Calorie (big ‘c’) is the amount of energy required to heat 1 kilogram (kg) of water 1 degree Celsius. You can do this by placing the apple in a special piece of equipment called a bomb calorimeter (image below), burn the apple, and measure how much heat transferred to the water. If the heat needed to burn the apple increased the temperature of the water by 100 degrees Celsius, viola (!) the apple contained 100 Calories.
A pedantic point on Calories, calories, and kilocaloriesp
“So where does ‘kcals’ come into play?” In an effort to read your mind, let me also address the difference between Calorie (big ‘c’), calorie (small ‘c’), and kilocalorie (kcal)—because there is a difference. One Calorie (i.e. 1 kilocalorie [kcal]) is equal to 1,000 calories (small ‘c’), just as one kilogram is equal to 1,000 grams [1]. Thus, we (the nutrition field) tend to use kcal rather than Calorie, but colloquially also throw around the term calorie (small ‘c’) despite it not being technically correct. From here on out, when I say ‘calorie’ I am referring to kcals as is customarily the case.
1 Calorie = 1 kcal = 1,000 calories
I will also note that some papers will use Joules (J) or kilojoules (kJ) as the measure of energy content of food. One Calorie is equal to 4.184 J for those who care—which is likely none of you. Okay, pedantic point over.
Nutrients vs. Food
For the most part, people do not think about their diet purely in terms of calories (although many certainly do). Rather, people eat diets comprised of foods, which in turn are comprised of nutrients that they themselves contain a certain caloric content. Thus, if we know the nutrient composition of the food we can determine its caloric value. The primary nutrients (i.e. the macronutrients) in foods and their energy content are:
· Carbohydrate – 4 kcals/gram
· Lipid – 9 kcals/gram
· Protein – 4 kcals/gram*
· Fiber - ~2 kcals/gram
And when applicable:
· Alcohol – 7 kcals/gram
*Note: protein, when burned in a bomb calorimeter actually gives a value closer to 5.4 kcals/gram; however, 1.4 kcals is subtracted to account for some the energy content getting lost in the urine as urea.
Also worth noting is that the above values are averages. In reality, there are tons of lipid species and proteins that all differ in their caloric content. Averages are easier to work with, however, and to get lost in that level of minutiae would be dizzying. Thus, food contains nutrients, and nutrients have a given caloric value depending on their chemical composition—and this is where things start to get fun! (Or annoying depending on who you are.)
Is a calorie a calorie?
If you want to raise the hackles of people in the nutrition field, throw this little puppy into the conversation. (I double dog dare you!) Like all nutrition-related issues, the answer is not black and white, and the sheer level of vitriol that occurs in the back-and-forth argumentation is primarily a function of the question being a terrible question to begin with. (I had a wonderful professor that once told a student, “If you want better answers, ask better questions.” Truer words were never spoken.)
From the literal perspective, a calorie is indeed a calorie—just as an inch is an inch. No way around it. However, that is not what people are getting at when they ask the question. (Yet they nevertheless expect you to be a mind reader.) The “is a calorie a calorie” question is typically rooted in the fact that the body does not metabolize all calories (i.e. the different macronutrients that have the caloric value) the same way. To which I say, no shit.
The problem is that this little fact tends to be taken hostage and then used to make the strawman argument that not all calories are the same, and that calories have nothing to do with changes in body weight, insulin is the devil, or that you should not consider calories or some such nonsense. This is terribly wrong (as we will discuss shortly); but, before I get ahead of myself, let us at least examine, briefly, the very true claim that not all calories are the same, despite consuming similar caloric values of the varying macronutrients.
Lost calories from alcohol (DO NOT try at home)
In probably the best (and most fun) example of ‘a calorie is not a calorie’ comes from a truly cool study by Charles Lieber and company in the early 1990’s [2]. (The title of the paper discussing the topic is literally “Do alcohol calories count?”)
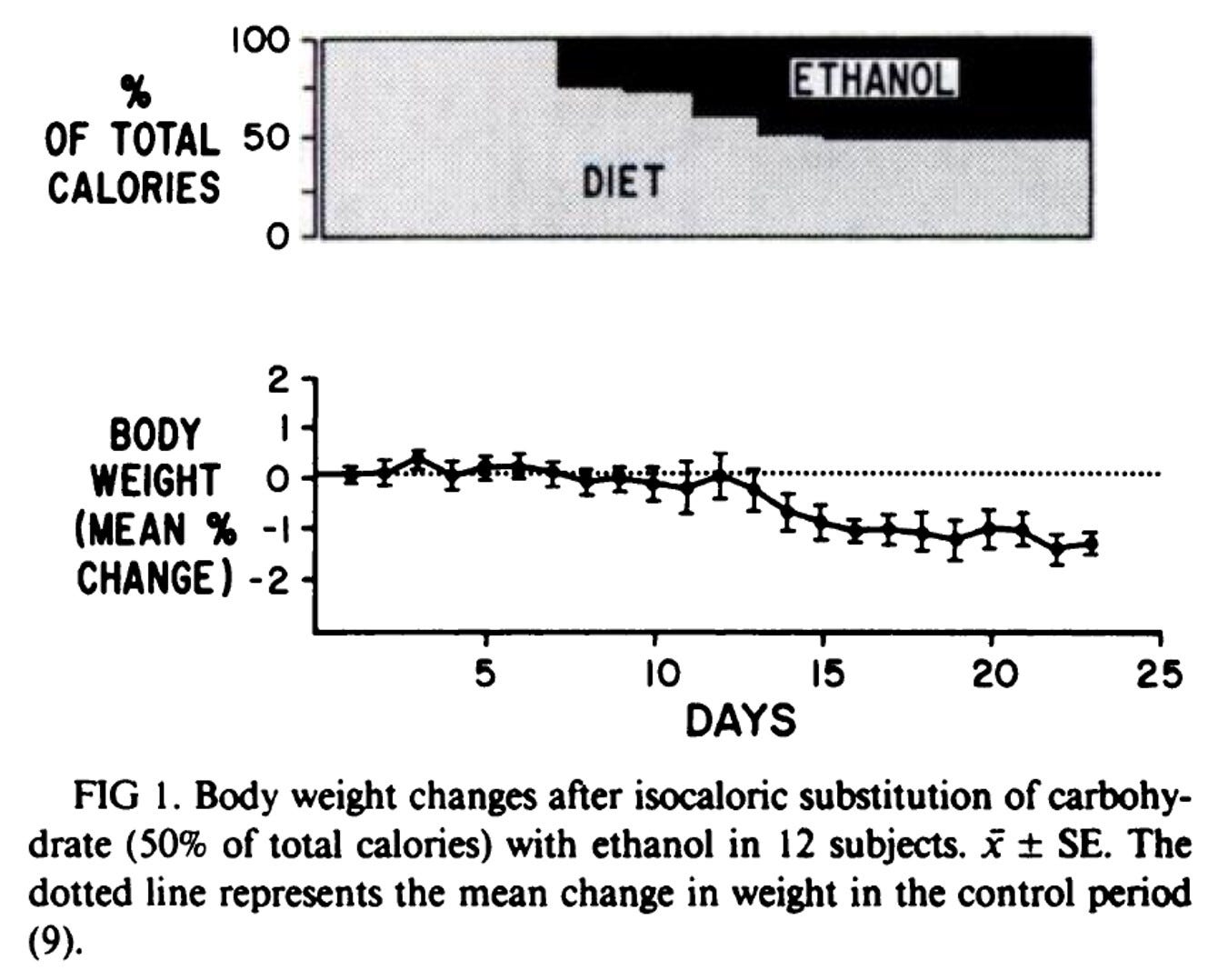
In this study, researchers took chronic alcoholics and recruited them to their metabolic ward where they gave them diets that varied in overall energy content (i.e. calories) as well as the composition of nutrients in the diet. For the first trial, each subject’s total calorie needs were measured, followed by a diet that met 100% of their calorie needs, but slowly replaced 50% of the calories with alcohol (remember, they were alcoholics and practice makes perfect). What happened, exactly? They lost weight!
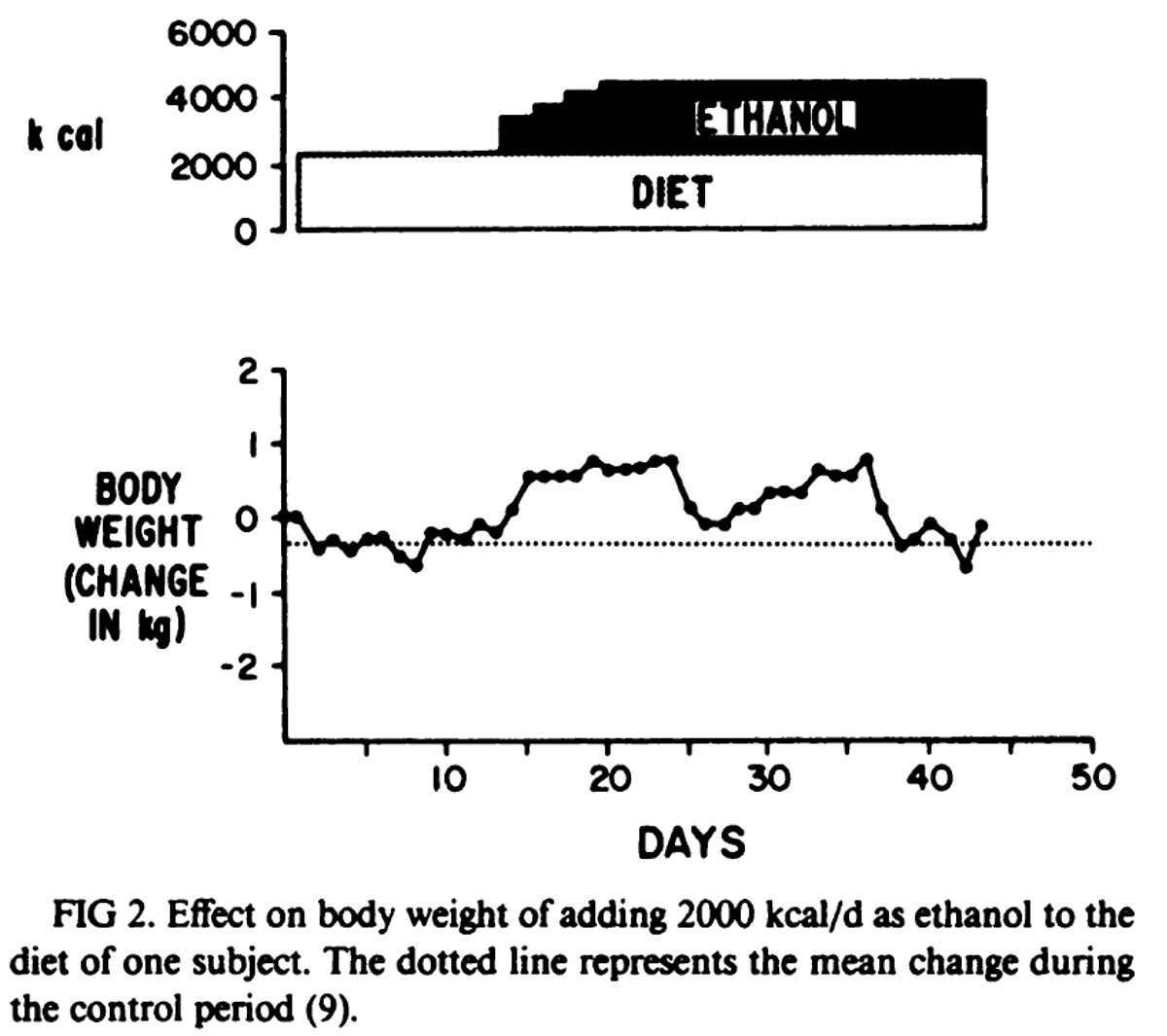
Next, the researchers returned whole food consumption to 100% of needs and then bumped up two of the subjects’ calories from ~2,500 calories to ~4,500 calories with the extra 2,000 calories coming purely from alcohol. In this condition, they ultimately maintained their weight!
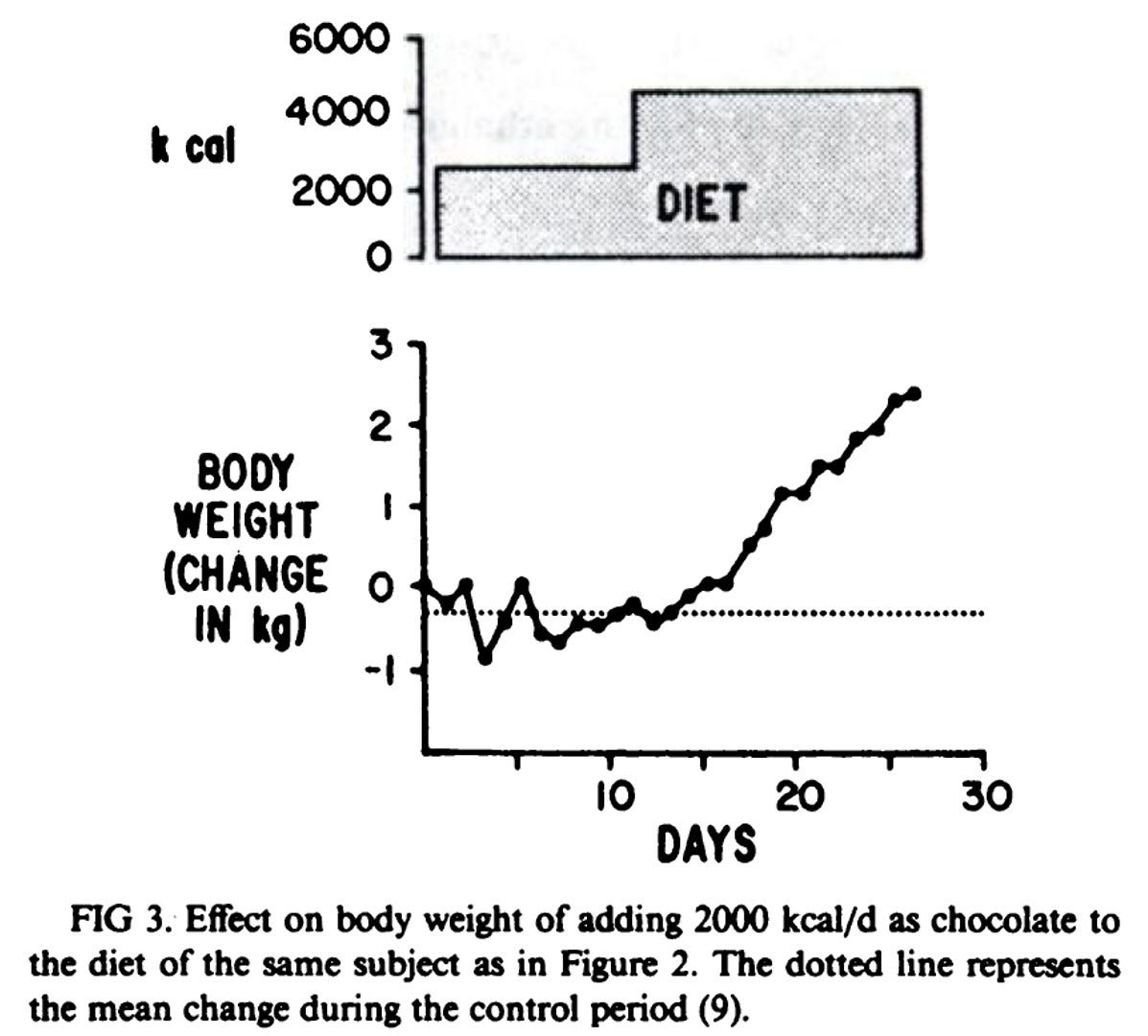
In the final condition, one remaining subject still consumed the 4,500 calories, but rather than alcohol comprising the excess 2,000 calories, the researchers replaced it with chocolate (what a study!). This time the subject rapidly gained weight!
As illustrated by this really neat study, it is obvious that not all calories (i.e. nutrients) are the same in terms of how the body metabolizes them and stores (or wastes) the excess energy. This goes for protein vs. carbs and fats and so on and so forth. No one worth his or her salt denies this fact. To the same extent, it does not also mean that calories do not matter. Quite the contrary! Remember, energy cannot be created or destroyed, only transferred. The question is always “where did the energy go?” In this instance, it turns out that it costs more energy to metabolize the alcohol in such chronic conditions.
(For those interested, intakes this chronically high seem to push an otherwise limited pathway called the MEOS pathway that, instead of producing energy from the breakdown of alcohol, actually consumes more energy in order to metabolize it. Other factors likely are involved and the entire story is not fully understood. Regardless, it’s still very interesting!)
Ultimately, all of this information points to the need to understand the framework under which changes in body weight/fat occur, and that is Energy Balance.
Energy balance
As we already covered, it is not uncommon to hear that calories do not matter. This, in my humble opinion, is absurd. Calories most certainly do matter when it comes to changes in body weight—which is not to say that this shit isn’t complicated. It just means that those who do not think that calories matter tend to not understand or disregard the governing framework for which changes in weight/fat mass occur—i.e. Energy Balance.
Sometimes referred to in common parlance as “calories in, calories out” (simply CICO), the Energy Balance Equation states that changes in a person’s weight (technically energy stores that make up that weight) are the mathematical difference between energy intake and energy expended [3].
Changes in energy stores = Energy in – Energy out
As we already pointed out, energy cannot be created or destroyed, only transferred. Therefore, if energy consumed is in excess of all expenditures (note how I phrased this, because this is where people have the most misunderstanding) then energy stores will increase and so will weight. Oppositely, if expenditures exceed intake, then energy stores will provide the difference and weight will decline. It is from this understanding that the common guidance of “eat less, move more” comes from. While true, it nevertheless misses the nuance and the intricacies of energy balance and appetite regulation that allow a person to actually achieve meaningful weight loss. Put another way, “eat less, move more” is easier said than done and more often than not is a useless piece of advice.
To complicate things even further, energy intake and expenditure are not completely separate sides of a coin but are intricately linked in a bidirectional manner—i.e. a change in one will lead to a concomitant change in the other. (This is where the magic happens!) Thus, whenever you decide to decrease energy intake or increase energy expenditure or store more energy in your body you will inevitably initiate a change in the other two. In order to better understand how this takes place, we first need to outline what constitutes energy intake and expenditure and how we measure these components. Then we can discuss how changes in one impact the other and interfere with changes in body weight/composition. We will begin with energy intake.
Energy intake
This is simply what you eat—i.e. food that is made up of nutrients that have a given caloric content. Well…almost. Just because you put food in your mouth, does not mean that it (the nutrients and their caloric content) actually get fully digested and absorbed into the body for utilization or storage. Indeed, if we look at the efficiency of fat, protein, and carbohydrate digestion and absorption, the numbers are quite variable, with fats having the highest absorptive efficiency at ~97% and proteins and carbs coming in much lower at ~80-95% depending on the source. (Not to mention any contributions from the gut microbiome, and digestive enzymes, etc. that are (re)absorbed.) Thus, energy intake is whatever is digested and absorbed minus what is lost in the feces and urine. After we account for the losses, whatever is left is metabolizable energy—i.e. it can be used to make ATP and do work, or is stored for later (as either glycogen or fat).
Metabolizable energy = Food energy – fecal/urinary energy losses
This, however, is not the end of the story. Just because that energy was absorbed and can now do something useful (like make ATP), we have to also layer on the fact that life is imperfect and that some of the energy will be wasted as heat due to metabolic inefficiencies. This is called the thermic effect of food (TEF; or diet-induced thermogenesis [DIT]). Typically averaged at ~10% of calories ingested, the metabolic cost of digesting, absorbing and metabolizing the macronutrients is also different, with protein winning the thermogenic advantage game. Moreover, the nature of the meal plays a role—i.e. whether it is comprised of whole/minimally processed foods or highly processed foods.
In a wonderful illustration, Barr and Wright [4] fed subjects two test meals: a whole grain, cheddar cheese sandwich or a processed white bread and processed cheese ‘product’ sandwich. Despite similar nutrient compositions and identical caloric content, the whole-food condition lead to a higher thermic effect than the processed meal. Put another way, more calories were expended to digest, absorb and metabolize the whole grain, cheddar cheese sandwich than the white bread, cheese ‘product’ sandwich.
In the end, whatever energy remains after all of this can support bodily demands or, if not needed, is stored for later. And while all of this is well and good, what has been excluded from the discussion thus far is how we actually measure energy intake in people—the punchline here being we have no good way to (accurately!) measure energy intake in free-living individuals. Therefore, unless you are measuring, preparing and feeding every gram of food/beverage to someone in an ultra-controlled setting, you are, at best, guessing what someone is actually eating in terms of energy content. And what are our ‘best guess’ methods?
Methods to measure energy intake
While it may seem trivial, estimating a free-living individuals’ energy intake is notoriously difficult. A handful of methods exist to inventory and calculate individuals’ energy intakes, but they are all flawed and terribly inaccurate. The most commonly used methods are the food frequency questionnaire (FFQ), food record, and a 24 h diet recall [5]. Modifications of these methods also exist—e.g. including newer digital technologies to improve self-reporting [6]. These versions, however, are not yet widely used and are beyond the scope of what I want to talk about here. Therefore, I will stick to the traditional ones listed above for the sake of brevity.
For the perspicacious reader, you noticed that I explicitly said “free-living” individual a couple of times already. This is to differentiate from the studies that do exist where subjects are admitted to metabolic wards entirely for days to weeks on-end and every single gram of food they receive is weighed and recorded and even fecal matter is subjected to bomb calorimetry to estimate fecal losses. (No guesswork there!) Sadly, I am not talking about those rare, beautiful, majestic trials.
FFQ
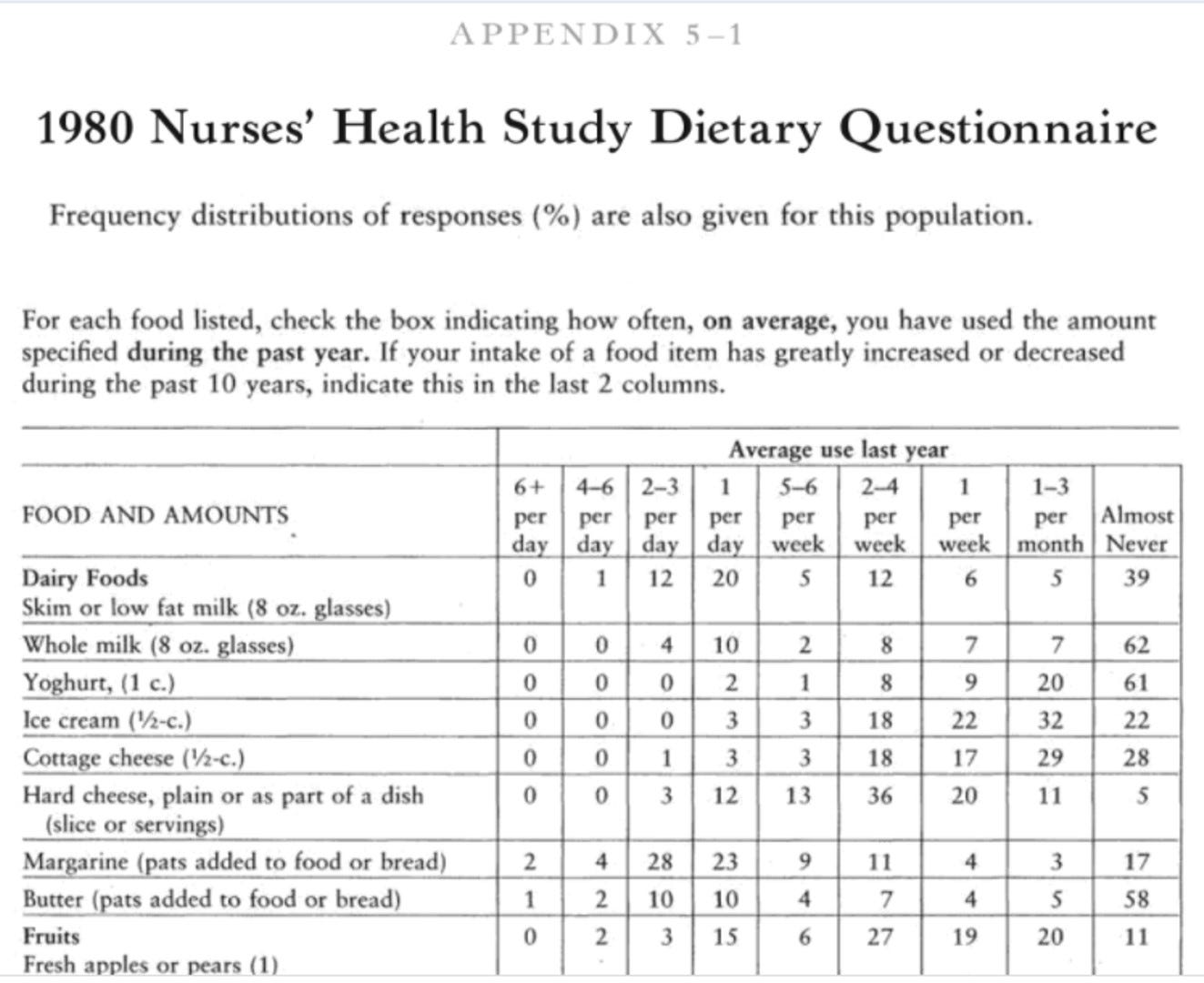
This method is exactly what it sounds like—it is a questionnaire that contains a whole host of foods with portion sizes and preparation methods that asks, on average (for the past month or year or so), how much you consumed of the following foods each week (see example below, taken from Ref [7]).
You can easily start to see the limitations present in this method. To name a few, outside of limitations of memory and honesty, if a food or preparation method is not present on the questionnaire, it will not be captured in the assessment. Some argue these limitations, among others, is why so many epidemiological studies (large-scale nutrition studies that assess hundreds if not thousands of people by using FFQs) lack any useful explanatory power [8]. Additionally, once the FFQ is completed someone needs to calculate overall intake using nutritional software, which also has its limitations based on how accurate the food selections are. If the software is out of date, you could be using an older, less accurate calculation of how many calories, etc. are present in a given food. A recent systematic review found that FFQs underestimate energy intake anywhere from 4.6 to 41% [9].
Food record
This method requires subjects to personally record all food and beverage intake to the best of their ability (which isn’t great for most) for a defined period of time (e.g. 3 days out of the week, typically 2 week days and 1 weekend day). Again, the limitations are not trivial—e.g. forgetting or not knowing to record something, willfully omitting certain foods to please the investigators, or outright changing your dietary habits during the study period. For those who do not rigorously think about their diet, asking them to complete these records is arduous and time consuming. Moreover, it is extremely easy to just not record your intake one day that your intake was poor and then record the next day when your diet was better. (Which begs the question, which is truly reflective of that person’s long-term intake?) Much has to be taken on faith with these records. To quote the aforementioned systematic review [9]:
“Of the studies that reported the accuracy of food records at the group level, the majority of studies (n=19) found significant underreporting of EI, by 11 to 41% […] with over-reporting found in only one study by 8% […] Three studies found no significant difference between absolute EI estimated by food record and TEE measured by DLW.”
24 h diet recall
This method is exactly as it sounds—ask the participant to recall everything he or she ate in the past 24 hours. Obviously, this is highly prone to errors in memory and honesty. Moreover, the person interrogating needs to know how to ask the right questions to uncover every ounce of food consumed (You had coffee? Great! How much? Was there sugar? Or possibly flavor pumps? How much? Do you use cream or milk? Milk. Okay, how much? Was it 1%? 2%? Whole? You get the picture.) Finally, how much does ONE DAY reflect anything about habitual diet?
Which method is best?
After examining the above, that leaves us with the question of which method is the best to estimate true dietary/energy intakes? Obviously the most accurate method is to have subjects reside in a metabolic ward and feed them every gram of food that the research team prepared and rigorously control their contact with visitors from outside—which has been done but is wildly expensive and not practical for most researchers/studies (particularly ones trying to understand dietary patterns and disease outcomes).
Ultimately, it comes down to the research question at hand and the level of error you are willing to tolerate. Obviously using a 24 h diet recall to represent long-term dietary intake is preposterous and a FFQ would be more suitable despite its limitations. Using multiple methods over time is probably best practice, coupled with some form of biomarker related to the nutrient or outcome in question—assuming a biomarker exists and is reliable. What is certain is that all these methods have been shown to be horribly inaccurate at estimating energy intake when compared to the doubly labeled water method (DLW) [9, 10]—a highly accurate, unbiased method to determine an individual’s total daily energy expenditure. Moreover, additional factors such as socioeconomic status, sex, and level of dietary restraint have been shown to affect dietary reporting [11, 12], thus further complicating the matter.
Summing up
To recap everything discussed thus far:
Energy is the capacity to do work
The Calorie is a unit of measure of energy
Food is made up of nutrients (carbohydrate, protein, fat, fiber, alcohol) that, depending on their chemical structure, have different caloric values
We transfer the energy from food to the bonds of ATP to do work in our bodies to stay alive
Not all calories are the same and are metabolized differently by the body with different efficiencies
Energy not needed immediately for biological work is stored for later (as fat or glycogen)
Changes in body weight (energy stores) is the mathematical difference between energy intake and energy expended—this is the concept of Energy Balance
Energy intake is the difference between nutrients consumed, digested and absorbed minus any losses in the feces/urine and metabolic inefficiencies
There are a handful of ways of measuring energy intake in free-living individuals, the common methods being the food frequency questionnaire, food record, and 24 h diet recall (which are all terribly inaccurate)
Having covered the Energy Intake (‘Calories In’) side of the Energy Balance Equation, in Part 2 we will cover Energy Expenditure (‘Calories Out’), how to measure it, and how changes in energy intake and energy expenditure lead to bidirectional changes in the other thus posing problems for weight loss and leading people to lose their ever-loving minds. (Or something to that effect.)
References
1. Hargrove, J.L., History of the calorie in nutrition. J Nutr, 2006. 136(12): p. 2957-61.
2. Lieber, C.S., Perspectives: do alcohol calories count? Am J Clin Nutr, 1991. 54(6): p. 976-82.
3. Schoeller, D.A., The energy balance equation: looking back and looking forward are two very different views. Nutr Rev, 2009. 67(5): p. 249-54.
4. Barr, S.B. and J.C. Wright, Postprandial energy expenditure in whole-food and processed-food meals: implications for daily energy expenditure. Food Nutr Res, 2010. 54.
5. Johnson, R.K., Dietary intake--how do we measure what people are really eating? Obes Res, 2002. 10 Suppl 1: p. 63S-68S.
6. Archundia Herrera, M.C. and C.B. Chan, Narrative Review of New Methods for Assessing Food and Energy Intake. Nutrients, 2018. 10(8).
7. Willett, W., Nutritional epidemiology. 2nd ed. Monographs in epidemiology and biostatistics. 1998, New York: Oxford University Press. xiv, 514 p.
8. Kristal, A.R., U. Peters, and J.D. Potter, Is it time to abandon the food frequency questionnaire? Cancer Epidemiol Biomarkers Prev, 2005. 14(12): p. 2826-8.
9. Burrows, T.L., et al., Validity of Dietary Assessment Methods When Compared to the Method of Doubly Labeled Water: A Systematic Review in Adults. Front Endocrinol (Lausanne), 2019. 10: p. 850.
10. Lissner, L., et al., OPEN about obesity: recovery biomarkers, dietary reporting errors and BMI. Int J Obes (Lond), 2007. 31(6): p. 956-61.
11. Hill, R.J. and P.S. Davies, The validity of self-reported energy intake as determined using the doubly labelled water technique. Br J Nutr, 2001. 85(4): p. 415-30.
12. Livingstone, M.B. and A.E. Black, Markers of the validity of reported energy intake. J Nutr, 2003. 133 Suppl 3: p. 895S-920S.